IRON
TRANSITION ELEMENT: IRON GROUP
| Atomic number: | 26 |
| Group numbers: | 8 |
| Period: | 4 |
| Electronic configuration: | [Ar] 3d6 4s2 |
| Formal oxidation number: | +2 +3 |
| Electronegativities: | 1.83 |
| Atomic radius / pm: | 124.1 |
| Relative atomic mass: | 55.845(2) |
Iron has been known since ancient times. The origin of the name comes from the Latin word ferrum meaning iron. It is a malleable, ferromagnetic, ductile, silvery-white metal. Exposed surfaces form red-brown oxides. Iron forms very strong alloys (steel). Iron dust flammable. It's the Fourth most abundant element in the earth's crust. Iron is obtained from iron ores. Pure metal is produced in blast furnaces by layering limestone, coke and iron ore and forcing hot gasses into the bottom. This heats the coke red hot and the iron is reduced from its oxides and liquefied where it flows to the bottom. Iron is the most common metal in human society. More than 90% of all metal refined in the world is iron. It is used in steel and other alloys. Iron is the chief constituent of hemoglobin which carries oxygen in blood vessels. It's oxides are used in magnetic tapes and disks. The price of 99.97 % pure iron pieces is 53.00 € for 1000 g.
| Density / g dm-3: | 7874 | (293 K) |
| 7035 | (m.p.) | |
| Molar volume / cm3mol-1: | 7.09 | (293 K) |
| 7.94 | (m.p.) | |
| Electrical resistivity / µΩcm: | 9.71 | (20 °C) |
| Thermal conductivity / W m-1K-1: | 80.2 |
| Melting point / °C: | 1538 |
| Boiling point / °C: | 2861 |
| Heat of fusion / kJ mol-1: | 14.9 |
| Heat of vaporization / kJ mol-1: | 340.2 |
| Heat of atomization / kJ mol-1: | 413.96 |
| First ionization energy / kJ mol-1: | 762.47 |
| Second ionization energy / kJ mol-1: | 1561.90 |
| Third ionization energy / kJ mol-1: | 2957.49 |
| in the atmosphere / ppm: | - |
| in the Earth's crust / ppm: | 70700 |
| in the oceans / ppm: | 0.01 |
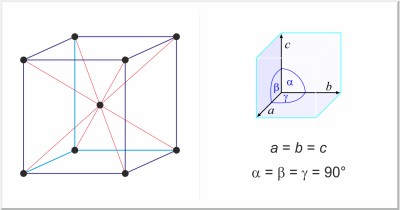
| Crystal structure: | body-centered cubic |
| Unit-cell dimensions / pm: | a=286.645 |
| Space group: | Im3m |
| Isotope | Relative atomic mass | Mass percent (%) |
|---|---|---|
| 54Fe | 53.939615(1) | 5.8(1) |
| 56Fe | 55.934942(1) | 91.72(30) |
| 57Fe | 56.935399(1) | 2.1(1) |
| 58Fe | 57.933280(1) | 0.28(1) |
| Balanced half-reaction | Eo / V | |
|---|---|---|
| Fe3+ + 3e- → Fe(s) | - 0.04 | |
| Fe3+ + e- → Fe2+ | +0.771 | |
| Fe3+ + e- → Fe2+ | +0.700 | (1 mol dm-3 HCl) |
| Fe3+ + e- → Fe2+ | +0.732 | (1 mol dm-3 HClO4) |
| Fe3+ + e- → Fe2+ | +0.68 | (1 mol dm-3 H2SO4) |
| Fe2+ + 2e- → Fe(s) | - 0.440 | |
| FeOH2+ + H+ + e- → Fe2+ + H2O | +0.91 | |
| Fe(OH)2+ + 2H+ + e- → Fe2+ + 2H2O | +1.19 | |
| FeO42- + 8H+ + 3e- → Fe3+ + 4H2O | +1.70 | |
| FeO42- + 7H+ + 3e- → FeOH2+ + 3H2O | +1.65 | |
| FeO42- + 6H+ + 3e- → Fe(OH)2+ + 2H2O | +1.56 | |
| FeO42- + 5H+ + 3e- → HFeO2 + 2H2O | +1.00 | |
| HFeO2- + 3H+ + 2e- → Fe(s) + 2H2O | +0.49 | |
| Fe(CN)63- + e- → Fe(CN)64- | +0.36 | |
| Fe(CN)63- + e- → Fe(CN)64- | +0.71 | (1 mol dm-3 HCl) |
| Fe(CN)63- + e- → Fe(CN)64- | +0.72 | (1 mol dm-3 HClO4) |
| Fe(CN)63- + e- → Fe(CN)64- | +0.72 | (1 mol dm-3 H2SO4) |
| Fe(C12H8N2)33+ + e- → Fe(C12H8N2)32+ | +1.06 |
| 25 Manganese | ← | 26 Iron | → | 27 Cobalt |
Citing this page:
Generalic, Eni. "Iron." EniG. Periodic Table of the Elements. KTF-Split, 13 Feb. 2025. Web. {Date of access}. <https://www.periodni.com/fe.html>.
Articles and tables
- Periodic table
- Online calculators
- Scientific calculator for chemists
- Gas laws calculator
- Molar mass calculator
- Angle converter
- Roman numerals converter
- Number systems converter
- Preparation of solutions
- Labeling of chemical containers
- Oxidation numbers calculator
- ARS method
- Oxidation number change method
- Ion-electron method
- Gauss elimination method
- Memory game
- Find the pairs
- Articles and tables
- Chemistry
- List of abbreviations and acronyms
- Crystal systems and Bravais lattices
- GHS - Hazard pictograms
- NFPA 704 Hazard Diamond
- Fundamental physical constants
- Solubility product constants
- SI - International System of Units
- Composition of mixtures and solutions
- Stoichiometric calculations
- Chlorinity and salinity of seawater
- Rare earth elements (REE)
- Ecology
- Web design
- Chemistry dictionary
- Chemistry
- Downloads
- ≡ Menu
